Research
The human brain of many individuals is genetically susceptible to neurodegenerative diseases like Alzheimer’s disease (AD) or poor recovery from traumatic injury and chemotherapy. However, pinpointing the genetic elements that promote this pathogenesis, establishing their underlying cellular and molecular mechanisms, and developing therapeutic strategies remain major challenges. These challenges are reflected in the growing patient population and complete absence of therapeutics for Alzheimer’s disease and related dementias. We need new approaches to understand the uniquely human biology that is fundamental to neurodegeneration and innovative technology that enables the development of effective therapeutic and preventative strategies. To fulfill this need, we engineer 3D models of human brain tissue from stem cells and apply these models to understand and therapeutically target risk factors for Alzheimer’s disease and other forms of cognitive impairment.
Our work specifically aims to (1) establish the cellular and molecular pathways underlying neurodegenerative risk factors, (2) target and modulate these pathways as potential therapeutic strategies, and (3) establish pioneering technology and tools that unravel complex genetic, cellular, and environmental interactions in the human brain. Ultimately, our goal is to facilitate a precision medicine approach to understanding and treating neurodegenerative disease.

Joel Blanchard, PhD
Associate Professor
Nash Family Department of Neuroscience
Department of Cell, Developmental & Regenerative Biology
Black Family Stem Cell Institute
Ronald M. Loeb Center for Alzheimer’s Disease
Icahn School of Medicine at Mount Sinai
Publications
2026
- Coccia, E. et al. ATP13A2 Loss of Function-Driven Polyamine Dysregulation Induces SAM Depletion and Epigenetic Astrocyte Toxicity. (2026) doi:10.64898/2026.04.02.716164. Link
- Jang, B. et al. A meta-analysis of single-nucleus expression quantitative trait loci linking genetic risk to brain disorders. Nature Genetics. 58, 737–747 (2026). Link
2025
- Schuldt, B. R. et al. APOE4 promotes cerebrovascular fibrosis and amyloid deposition via a pericyte-to-myofibroblast transition. bioRxiv 2025.09.04.674192 (2025) doi:10.1101/2025.09.04.674192. Link
-
Bright, A., Akay, L. A., Blanchard, J. & Tsai, L.-H. Emerging connections between myelin and Alzheimer’s disease. Nature Cell Biology. 27, 1049–1051 (2025). Link
-
Mesentier-Louro, L. A. et al. Cholesterol-mediated Lysosomal Dysfunction in APOE4 Astrocytes Promotes α-Synuclein Pathology in Human Brain Tissue. (2025) doi:10.1101/2025.02.09.637107. Link
- Goldman, C. et al. Genetically encoded and modular subcellular organelle probes reveal dysfunction in lysosomes and mitochondria driven by PRKN knockout. iScience 28, 112816 (2025). Link
- Morrone Parfitt, G., Coccia, E., Goldman, C. et al. Disruption of lysosomal proteolysis in astrocytes facilitates midbrain organoid proteostasis failure in an early-onset Parkinson’s disease model. Nat Commun 15, 447 (2024). Link
2024
- Qian Wang et al. Molecular profiling of human substantia nigra identifies diverse neuron types associated with vulnerability in Parkinson’s disease. Sci. Adv.10,eadi8287(2024) Link
- Sarrafha L., Neavin, DR. Parfitt, GM. Kruglikov, IA. Whitney, K. Reyes, R. Coccia, E. Kareva, T. Goldman, C. Tipon Regine, T. Croft, G. Crary, JF. Powell, J. Blanchard J.* Ahfeldt, T*., Novel human pluripotent stem cell-derived hypothalamus organoids demonstrate cellular diversity. iScience (2023) in press.Link
2023
- Louro, L. Suhy, N. Broekaart, D. Bula, M. Pereira AC. Blanchard, J. Modeling the Blood-brain barrier using Human induced pluripotent stem cells. Methods Mol Biol. (2023), PMID: 37300772Link
- Goldman, G., Suhy, N., Louro, L., Rooklin, R., Blanchard J., Reconstruction of the blood-brain barrier in vitro to model and therapeutically target neurological disease. JoVe (2023). Link
2022 – 2018
- Blanchard J., Akay, LA., Velderrian JD., von Maydell, D., Mathys, H., Davidson SM., Effenberger, A., Chen C., Maner-Smith, K., Hajjar, I., Ortlund, EA., Bula, M., Agbas, E., Ng, A., Jiang, X., Kahn, M., Blanco C., Laoie N., Liu L., Reyes, R., Lin YT., Ko T., R’Bibo LR., Ralvenius WT., Bennett DA., Cam HP., Kellis M., Tsai L-H. APOE4 impairs myelination via cholesterol dysregulation in oligodendrocytes. Nature (2022) link
- Blanchard J., Blenkinsop T., Ghaffari S., Krauss RS., Rendl M., Sturgeon CM., Zwaka TP., Marek ., Millar S, Fulfilling the promise of stem cell biology and regenerative medicine. Science The Frontiers of Medical Research (2021) Link
- Blanchard J., Victor M., Tsai L-H, Dissecting the complexities of Alzheimer’s disease with in vitro models of the human brain. Nature Reviews Neurology (2021). Link
- Blanchard J., Bula M., Akay, LA., Zhu L., Frank A., Velderrian JD., Victor, M., Bonner, JM., Mathys, H., Lin YT., Kellis, M., Cam, H., Tsai LH. Reconstruction of the human blood-brain barrier in vitro reveals a pathogenic mechanism of APOE4 in pericytes. Nature Medicine (2020)Link
- Blanchard J., Tsai, LH., Unraveling the paradox of statins with human neurons: New leads in Alzheimer’s disease. Cell Stem Cell (2019). Link
2018 – 2009
- Tsunemoto R.*, Lee S.*, Szűcs A., Chubukov P., Sokolova I., Blanchard J., Eade K., Bruggemann J., Su A., Sanna P., Baldwin K., Binary reprogramming codes for neuronal identity and diversity. Nature (2018). Link
- Blanchard J., Xie, J., El-Mecharrafie, N., Lerner, RA., Baldwin K., Replacing reprogramming factors with antibodies selected from combinatorial antibody libraries. Nature Biotechnology (2017). Link
- Blanchard J*., Eade K*., Szucs A., Lo Sardo V., Tsunemoto R., Williams D., Sanna P., Baldwin K. Selective conversion of fibroblasts into peripheral sensory neurons. Nature Neuroscience (2015) Link
- Tsunemoto R., Eade, K., Blanchard J., Baldwin K. Forward engineering neuronal diversity using direct reprogramming. EMBO J (2015). Link
- Wang Y., Davidow L., Arvanites A., Blanchard J., Lam K., Woo J., Coy S., Rubin L.L., McMahon A. Selective Identification of Hedgehog pathway antagonists by direct analysis of Smoothened Ciliary Translocation. ACS Chemical Biology (2012). Link
- Wang Y., Davidow L., Arvanites A., Blanchard J., Lam K., Woo J., Ng J., Curran T., Rubin L.L., McMahon, A. Glucocorticoids Compounds Modify Smoothened Localization and Hedgehog Pathway Activity. Chemistry and Biology. (2012). Link
- Ichida J. K*., Blanchard J*., Lam K*., Son E. Y*., Chung J. E., Egli D., Loh K. M., Carter A. C., Di Giorgio F. P., Koszka K., Huangfu D., Akutsu H., Liu DR., Rubin LL., Eggan A Small-Molecule Inhibitor of Tgf-β Signaling Replaces Sox2 in Reprogramming by Inducing Nanog. Cell Stem Cell (2009). Link
*These authors contributed equally to this work
Lab Members

Louise Louro, PhD
Instructor
Louise has a background in central nervous system injury, biomarkers and regenerative strategies, including cell therapies and pharmacological approaches. Her current research is focused on developing multi-cellular in vitro models of neurodegenerative diseases. She enjoys cooking Brazilian-Italian food and walking around the city to find nice little wine and food spots.

Ashley Harlock
PhD Student
Ashley graduated from Michigan State University in 2024 with Bachelor’s degrees in Neuroscience and Human Biology. During undergrad, she explored a wide range of research—from memory and addiction to particle physics—and led an independent project on how cocaine affects endoplasmic reticulum stress in the brain. She also played Division I field hockey at MSU, where she was a three-year team captain and juggled training with a full academic and research load. Now a PhD student in Neuroscience at the Icahn School of Medicine at Mount Sinai, Ashley studies traumatic brain injury and Alzheimer’s disease using 3D stem cell-derived models of human brain tissue in the Blanchard Lab. Outside the lab, she’s a member of the NYC Field Hockey Club and loves singing, playing guitar, and being the ultimate foodie around New York City.

Anna Bright
Doctoral Student
Anna graduated from Vanderbilt University in 2022 with a Bachelor’s degree in neuroscience. Her undergraduate research in Dr. Vivian Gama’s laboratory focused on mitochondrial biology in neurodevelopment. Anna’s current research interests involve using 3D stem cell-derived models to better understand the biological mechanisms of Alzheimer’s disease. When she’s not in lab, you can find Anna painting, thrifting, or roaming around Central Park admiring the dogs.

Julia Gesner
Masters Graduate Student
Julia graduated from Lafayette College in 2024 with a B.S. in Neuroscience and Honors in Psychology, magna cum laude. As an undergraduate, she studied the gene CNTNAP2, implicated in autism and dyslexia, using mouse behavioral assays, and completed a senior thesis on the effects of stress and self-efficacy on test anxiety. She is currently investigating polyamine dysregulation in Alzheimer’s and Parkinson’s disease using APOE3 and APOE4 astrocytes. Outside the lab, Julia is a certified EMT and medical scribe, currently applying to medical school with aspirations of becoming a physician. In her free time, Julia enjoys spending time with friends and family, baking and napping.

Ana Forton-Juarez
Doctoral Student

Tatyana Kareva
Lab Manager
Tatyana obtained her B.S in Medical Laboratory Science from City Medical College in Odessa, Ukraine. She is supporting an administrative as well as technical side of the research projects in the lab. In her free time , Tatyana enjoys exploring New York City and spending time with family.
![Andrea Headshot[85]](https://labs.neuroscience.mssm.edu/wp-content/uploads/2023/11/Andrea-Headshot85-150x150.jpg)


Andrea Perez Arevalo, PhD
Post Doctoral Fellow
Andrea obtained her PhD in Human Biology from Ulm University, Germany, in 2023. Her research in Prof. Dr. Tobias M. Böckers lab focused on understanding the role of microglial cells in the development of Autism Spectrum Disorders (ASDs). Her current research is focused on understanding the influence of glial cells in the development of neurodegenerative diseases in different genetic variants and environmental conditions. Outside the lab you can find Andrea enjoying music and art around the city, trying new foods, and spending time in nature.
Natalie Suhy
Doctoral Student
Natalie graduated from UC Berkeley in 2021, where she worked in the lab of Dr. David Schaffer and received a Bachelor’s degree in molecular and cell biology. Her background is in stem cell biology, CRISPR gene editing, and 3D pluripotent stem cell cultures for use in cell replacement therapy. She also has prior research experience studying cancer immunology with Merck, and gene therapy techniques with biotech startups. Natalie’s current research interests include modeling of the blood brain barrier, examining genetic components and identifying immunological mechanisms of neurodegenerative diseases. In her free time, she enjoys cooking vegan recipes, going out with friends, and trying new workout classes.
Sebastian Gaese
Associate Researcher
Sebastian graduated from Kenyon College in 2022 with a Bachelor’s degree in Neuroscience. As an undergraduate, he worked in Dr. Sarah Petersen’s laboratory studying neurodevelopmental changes in zebrafish. Following graduation, Sebastian joined the Neuroregeneration Institute at McLean Hospital, where he worked on an autologous iPSC-derived dopamine neuron cell therapy for Parkinson’s disease patients and examined the effects of APOE on glial and inflammatory responses in stem cell and murine models. Sebastian is currently interested in using iPSC-derived cells to study neurodegeneration and developing novel technologies to advance brain research.

Elena Coccia, PhD
Instructor
Elena obtained her Ph.D. in Neuroscience from University Autonoma of Barcelona in 2020. She is focused on understanding cell type-specific effects of Parkinson’s Disease genetic variants in pluripotent stem cells models. Outside of the lab, Elena loves to explore NYC, travel and try new food.

Braxton Schuldt
MD/PhD Student
Braxton graduated from Amherst College in 2021 with a Bachelor’s degree in neuroscience. For his undergraduate thesis, Braxton studied the molecular mechanisms of dendritic spine development in the context of autism spectrum disorder. He is currently interested in using stem cell models to study neurodegeneration and explore the potential of neurosurgical transplantation as a therapeutic intervention for CNS diseases. Outside of the lab, Braxton enjoys running, hanging out with friends, and eating Domino’s pizza

Georgia Gallagher
Associate Researcher
Georgia graduated from Bowdoin College with a bachelor’s degree in Neuroscience in May 2025. During her undergraduate years, she studied in William Jackman’s lab studying genetic differences in Fgf10 expression in zebrafish and as a fertility specialist intern studying reproductive cell development. She later joined Columbia University as a research intern, where she engineered plasmids to measure minimally invasive gCAMP luciferase readouts in both neural stem cells and mouse models. Georgia is interested in neuroinflammation, the role of immunology role in neurodegeneration, and regenerative medicine. She enjoys cooking, spending time with friends and exploring the new museums/ neighborhoods in the city.



Youbin Kim, PhD
Post Doctoral Fellow
Youbin obtained her Ph.D. in Neuroscience from Seoul National University in 2023. Her research in Dr. Yong-Keun Jung’s lab focused on identifying receptors involved in the spread of tau between neurons in tauopathies. She is currently studying lipid metabolism in glial cells in relation to Alzheimer’s disease. In her free time, Youbin enjoys discovering new music and exploring New York City.
Abigail Uchitelev
Undergraduate Researcher
Abigail is an undergraduate student at the Macaulay Honors College at Hunter College pursuing a Bachelor’s degree in Human Biology with the goal of becoming a physician after completing medical school. She is now delving into her strong passion for understanding the intricacies of the human brain as an intern at the Blanchard lab, working with in vitro models of neurodegenerative diseases. Her current research focuses on the retinal pigment epithelium and how it connects to the blood-brain barrier. Abigail aims to contribute to the scientific and medical field by pursuing her interests in regenerative medicine and cellular biology. In her free time, she enjoys spending time with her family, hiking, and experimenting with new recipes.
Miranda Yang, PhD
Assistant Professor



Jennifer Strong
Doctoral Student
I’m interested in 3D stem cell models to explore Neurodegeneration and aging, specifically in the context of Alzheimer’s, Parkinson’s, & ALS
Alice Buonfiglioli, PhD
Instructor
Alice obtained her Ph.D. in Neuroscience from Charité University of Medicine in collaboration with the Max-Delbrück-Center for Molecular Medicine in Berlin, Germany, in 2020. Her graduate and postgraduate research has focused on investigating microglia in physiology and pathology in murine and human stem cell-derived 2D and 3D models. She is currently studying genetic variants in microglia in the context of neurodegeneration using iPSC technology. Outside the lab, Alice plays volleyball, enjoys music and traveling.
Dominic Haworth-staines, MS
Research Associate
Dominic is a recent MSc graduate from Imperial College London. For his thesis, he worked in Dr. Sonia Gandhi’s lab studying the toxicity of alpha-synuclein oligomers in cortical neurons. He is particularly interested in regenerative medicine and stem cell biology to model neurodegenerative disease. Outside of the lab, Dominic likes to spend time with friends, walk around the city and explore cheap eats in Chinatown.
Alumni
| Ricardo Reyes | Harvard Medical School, MD Student |
| Emily Sartori | Icahn School of Medicine, PhD Student |
| Sasha Frank | Icahn School of Medicine, MD/PhD Student |
| Lily Sarrafha, PhD | CalicoTherapeutics, Scientist |
| Camille Goldman, PhD | Oxford University, Postdoc |
| Jessica Schwarz | ZS, Consultant |
| Gustavo Parfitt, PhD | University College London, Faculty |
| Rikki Rooklin | University of Pittsburgh, PhD student |
Projects
Areas of Investigation
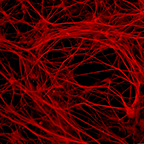
Blood-Brain Barrier (BBB)
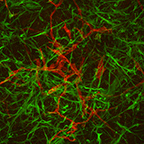
The BBB feeds the brain with nutrients and oxygen while also protecting it from peripheral pathogens and insults. It is increasingly recognized the BBB also has a prominent role in neurodegenerative disease. Numerous genes associated with risk for neurodegeneratoion are expressed in the cells that constitute the BBB where they likely directly influence disease susceptibility and pathogenesis. For example, APOE is highly expressed In the BBB and the APOE4 variants is known to correlate with increased amyloid deposition on cerebral vasculature. In addition to being a location of amyloid deposition, the BBB also forms a highly selective barrier that prevents more than 98% of all small molecules from entering the brain. By developing an iPSC-based model of the BBB, we aim to understand i) how the BBB contributes to neuorodegeneration susceptibility and pathogenesis and ii) develop a physiologically reliable test to asses drug-brain penetration across diverse genetic populations.
Myelination
It is emerging that oligodendrocytes (OLs) and myelin degeneration play a central role in neurodegeneration and cognitive aging. In the human brain, the total myelin volume increases until around the fourth or fifth decade of life after which it sharply declines. Therefore, myelin degeneration is thought to be a fundamental mechanism leading to cognitive changes associated with aging. In most neurodegenerative diseases, myelin degeneration is accelerated. For example, the majority of Alzheimer’s disease (AD) patients exhibit deficits in white matter compared to healthy individuals of the same age. Surprisingly, in populations at high risk for developing AD, several imaging studies identified that the loss of white matter begins prior to cognitive decline. Likewise, in rodent AD models, myelin deficits are observed prior to the formation of amyloid plaques and neurofibrillary tau tangles, the classical pathological hallmarks of AD. At the molecular level, many of the genes implicated in the development of sporadic AD could directly influence OL biology and/or myelination. Collectively, this suggests that myelin degeneration may be one of the earliest biomarkers and a key pathological mechanism underlying cognitive decline in AD and aging. Despite these implications the role of OLs and myelin degeneration in AD pathogenesis remain largely unclear. This is likely due to species differences in myelin biology, difficulty in procuring mature OLs, and limited assays for human myelination. Through developing technology to study human myelination in a dish we aim to gain deeper insight into how myelination is altered during neurodegeneration and the effect of genetics and the environment on these processes.
Neuro-Immune-Vascular Brain
In the human brain, neurons, microglia, and the blood-brain barrier are dynamically interconnected. How these connections are formed during development, maintained in homeostasis, and degenerate with age remain poorly understood. It is also largely unclear how human genetic diversity alters neuro-immune-vascular interactions to influence susceptibility to disease and age-related cognitive impairments. To approach these questions, we have developed an in vitro Neuro-Immune-Vascular tissue.
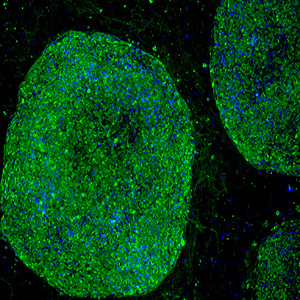
Multi-cellular integrated brain tissue (miBRAIN)
Numerous preclinical drugs have effectively cured Alzheimer’s disease in mice and other vertebrate models. However, all of these once-promising candidates have ultimately failed to show efficacy in human clinical trials. There are general concerns about the fidelity of rodent models and most of the genetic variants associated with increased risk for Alzheimer’s disease in humans are not conserved between species. Therefore, intermediate models that exhibits AD pathology in human tissues may better predict clinical outcomes. To address this, we are creating a multicellular integrated Brain tissue (miBRAIN) composed of seven disease-relavent cell-types. Future studies will apply this models to 1) pinpoint cellular and molecular mechanisms that promote risk for age-related cognitive impairment 2) screen and stratify drug response and efficacy across diverse genetic backgrounds 3) investigate how adverse life events such as traumatic brain injury or chemotherapy increase susceptibility to age-related neurodegeneration.
Awards
2021 Friedman Brain Institute Scholar Award
2019 – ISSCR Merit Award
2019 – ISSCR Travel Award, International Society for Stem Cell Research meeting, Los Angles, CA.
2018 – Postdoctoral Fellowship in Aging Research, Glenn Foundation of Medical Research
2014 – Betty Jean Ogawa Memorial Poster Award, International Society for Stem Cell Research
2014 – ISSCR Travel Award, International Society for Stem Cell Research meeting Vancouver, BC.
2014 – Keystone Scholarship, Keystone symposia on Stem cells and Reprogramming, Olympic Valley, CA.
2011 – Pre-doctoral Fellowship, California Institute of Regenerative Medicine.
2010 – ISSCR Travel Award, International Society for Stem Cell Research meeting San Francisco, CA.
2010 – Biotechnology Master’s Thesis Honorable Mention, Harvard University
2009 – ISSCR Travel Award, International Society for Stem Cell Research meeting, Barcelona Spain.
Lab News

Congrats to Jessica for wining the Alzheimer’s Association/NACC New Investigator Award! link
 Gustavo’s DJ1 paper published in Nature Communications! link
Gustavo’s DJ1 paper published in Nature Communications! link
Lily’s hypothalamic organoid paper was published in iScience! link

Lab receives grant to develop models of TBI using iPSC-derived human brain tissue link
 Elahi, Blanchard, and Raj team awarded Grant from CZI link
Elahi, Blanchard, and Raj team awarded Grant from CZI link
APOE4 myelin paper published in Nature
 Camille wins a NRSA F31 predoctoral fellowship!
Camille wins a NRSA F31 predoctoral fellowship!
 Louise wins ISSCR travel award and ISSCR Merit award!
Louise wins ISSCR travel award and ISSCR Merit award!
 Lab awarded a grant from NASA link
Lab awarded a grant from NASA link
 Lab awarded a grant from the Cure Alzheimer’s Fund link
Lab awarded a grant from the Cure Alzheimer’s Fund link

Nature Reviews Neurology article featured on Cover link
 Cell Stem Cell Profiles of Early-Career Researchers in the Time of COVID-19
Cell Stem Cell Profiles of Early-Career Researchers in the Time of COVID-19Funding
Active
Extending the longevity of human brain models for longitudinal analysis
Active
Establishing the molecular and cellular mechanisms and biomarkers of APOE4-mediated susceptibility to tau-related cognitive impairments
Studies have demonstrated that the strongest risk factor for Alzheimer’s disease APOE4 increases the severity of Tau pathology and cognitive impairments, but the mechanisms are unknown. Using our in vitro brain technology, we will dissect the cellular and molecular mechanisms by which APOE genotype modulates Tau phenotypes. This will reveal signaling pathways that we will investigate as therapeutic targets using chemical and genetic approaches.
Implications of Polyamine and Glucosylceramide Transport for Parkinson’s Disease (IMPACT-PD)
This grant seeks to understand the role of ATP13A2 and ATP10B in PD-associated lysosomal dysfunction. Our group will collaborate with other investigators in establishing, validating and characterizing all in vitro cell models. We will lead the efforts to generate midbrain dopaminergic neurons, midbrain patterned astrocytes, co-cultures and phenotyping efforts in hPSC derived differentiated cells.
UG3-NS115064 Construction of an integrated immune-vascular brain- chip as a platform for the study, drug screening, and treatments of Alzheimer’s disease
This grant builds off our iBBB technology by incorporating additional cells types to generate a human multicellular integrated brain tissue (miBrain). Using iPSC lines from which we have matched post-mortem brains we will develop, optimize, and validate this human brain-on-a-chip technology as platform for the study, screening and therapeutic optimization of Alzheimer’s disease.
R01NS114239 Understanding cell-type vulnerability and oxidative stress pathology in Parkinson’s Disease using isogenic human dopaminergic neurons
This grant investigates site-specific vulnerability of dopaminergic (DA) neurons in Parkinson’s Disease (PD) using hiPSC and organoid models. The mechanism of site-specific DA neuron vulnerability in PD is not yet known; insight into these mechanisms will uncover new therapeutic opportunities in PD.
Completed
UG3-NS115064S1 Determine vascular cell type-specific susceptibility to SARS-COV-2 and molecular response to the virus in vasculature microphysiological model
This grant supported by supplemental funding through NCATs allowed us to apply our miBrain technoloyg to Determine vascular cell type-specific susceptibility to SARS-COV-2 and molecular response to the virus in vasculature microphysiological model and evaluate effect of current treatment options and combinations on vasculature in microphysiological model via monitoring markers for thrombosis and vascular risk factors
Job opportunities