Research
The Maze laboratory is focused on understanding the complex interplay between chromatin regulatory mechanisms in brain and neuronal plasticity. We combine a wide variety of biochemical, biophysical, molecular and behavioral approaches to assess the role of histone proteins, and their associated posttranslational modifications, turnover dynamics and remodeling activities, in the regulation of normal neurodevelopment, and the contribution of such processes, or aberrations thereof, to neurological and psychiatric disease. We place particular emphasis on psychiatric disorders associated with monoaminergic (e.g., serotonin, dopamine, etc.) dysfunction, such as major depressive disorder and drug addiction, as well as neurodevelopmental syndromes resulting in deficits in synaptic plasticity and intellectual disability (e.g., Down syndrome and autism). Being both ‘basic’ and translational in nature, our work – so-called “mechanistic neuroepigenetics” – employs state-of-the-art gene and chromatin arraying technologies, analytical chemistry (e.g., mass spectrometry and 14C bomb pulse labeling) and neurobiological phenotyping, in both rodents and humans (postmortem brain and iPSC derived neurons), to explore novel chromatin phenomena in the central nervous system. This includes the identification and characterization of neuronally enriched histone modifications, chromatin “readers,” “writers” and “erasers,” all of which play critical roles in neuronal plasticity and behavioral adaptation.
![]()

Ian Maze, PhD
Howard Hughes Medical Institute Investigator
Professor, Neuroscience
Professor, Pharmacological Sciences
Director, Center for Neural Epigenome Engineering
Location
Lab: HESS CSM 9-201 / 10-302
Office: HESS CSM 9-109
Phone
Office: 212.824.8979
Lab: 212.824.9158 / 212.824.9172
Email
Connect with us!![]() @themazelab
@themazelab
Publications
2026
Reimagining biogenic amine signaling in the brain and beyond.
Vinson DA, Maze I.
Trends Neurosci. 2026 Jan;49(1):35-48. doi: 10.1016/j.tins.2025.11.003. Epub 2025 Dec 11. PMID: 41387034
Transcriptional regulation of ventral hippocampus-nucleus accumbens circuit excitability drives cocaine seeking.
Eagle AL, Sugimoto C, Doyle MA, Anderson D, Mousavi SL, Dykstra MM, Kuhn HM, Murray BR, Bastle RM, Simmons S, He J, Maze I, Mazei-Robison MS, Robison AJ.
Sci Adv. 2026 Mar 6;12(10):eadv1236. doi: 10.1126/sciadv.adv1236. Epub 2026 Mar 4. PMID: 41779851
Quinone reductase 2 reads H3 serotonylation to support neuronal maturation.
Chen M, Yang C, Li X, Kong L, Weekley BH, Wei X, O’Chan JC, Vinson DA, Cetin B, Ramakrishnan A, Shen L, Zeng R, Liu Z, Zhang J, Cappuccio KM, Sokol JR, Baljinnyam E, Hu R, Rosenblum K, Molina H, Zheng Q, David Y, Marro SG, Muir TW, Li XD, Li H, Maze I.
bioRxiv [Preprint]. 2026 Mar 19:2026.03.17.712426. doi: 10.64898/2026.03.17.712426. PMID: 41890023
Dopamine drives persistent remodelling of the maternal brain.
O’Chan JC, Di Salvo G, Cunningham AM, Dutta S, Brindley E, Weekley BH, Chen W, Iyer RR, Wan E, Zhang C, Mechawar N, Turecki G, Maze I.
Nature. 2026 May 20. doi: 10.1038/s41586-026-10509-4. Online ahead of print. PMID: 42162419
2025
Major-depressive-disorder-associated dysregulation of ZBTB7A in orbitofrontal cortex promotes astrocyte-mediated stress susceptibility.
Fulton SL, Bendl J, Di Salvo G, Fullard JF, Al-Kachak A, Lepack AE, Stewart AF, Singh S, Poller WF, Bastle RM, Hauberg ME, Fakira AK, Patel V, Chen M, Durand-de Cuttoli R, Gameiro-Ros I, Cathomas F, Ramakrishnan A, Gleason K, Shen L, Tamminga CA, Milosevic A, Russo SJ, Swirski FK, Slesinger PA, Abdus-Saboor I, Blitzer RD, Roussos P, Maze I.
Neuron. 2025 Aug 20;113(16):2656-2672.e13. doi: 10.1016/j.neuron.2025.05.023.
The SWI/SNF-related protein SMARCA3 is a histone H3K23 ubiquitin ligase that regulates H3K9me3 in cancer.
Akano I, Hebert JM, Tiedemann RL, Gao Q, Xiao Y, Prescott NA, Liu Y, Tan KS, Bastle RM, Ramakrishnan A, Maze I, Sidoli S, Koche RP, Ganesh K, Rothbart SB, David Y.
Mol Cell. 2025 Aug 7;85(15):2885-2899.e8. doi: 10.1016/j.molcel.2025.06.020. Epub 2025 Jul 17.
A Novel Role for the Histone Demethylase JMJD3 in Mediating Heroin-Induced Relapse-Like Behaviors.
Mitra S, Werner CT, Shwani T, Lopez AG, Federico D, Higdon K, Li X, Gobira PH, Thomas SA, Martin JA, An C, Chandra R, Maze I, Neve R, Lobo MK, Gancarz AM, Dietz DM.
Biol Psychiatry. 2025 Mar 15;97(6):602-613. doi: 10.1016/j.biopsych.2024.06.028. Epub 2024 Jul 15.
Early-life stress alters chromatin modifications in VTA to prime stress sensitivity
Luke T Geiger, Julie-Anne Rodier Balouek, Mason R Barrett, Jeremy M Thompson, Lisa Z Fang, Lorna A Farrelly, Andy S Chen, Megan Tang, Shannon N Bennett, Benjamin A Garcia, Ian Maze, Meaghan Claire Creed, Catherine Jensen Peña
bioRxiv [Preprint]. 2025 Apr 1:2024.03.14.584631. doi: 10.1101/2024.03.14.584631.
Persistent dopamine-dependent remodeling of the neural transcriptome in response to pregnancy and postpartum
Jennifer C Chan, Giuseppina Di Salvo, Ashley M Cunningham, Sohini Dutta, Elizabeth A Brindley, Ethan Wan, Cindy Zhang, Ian Maze
bioRxiv [Preprint]. 2025 Feb 25:2025.02.20.639313. doi: 10.1101/2025.02.20.639313.
Elucidating neuroepigenetic mechanisms to inform targeted therapeutics for brain disorders
Benjamin H. Weekley, Newaz I. Ahmed, Ian Maze
iScience. 2025 Feb 22;28(3):112092. doi: 10.1016/j.isci.2025.112092
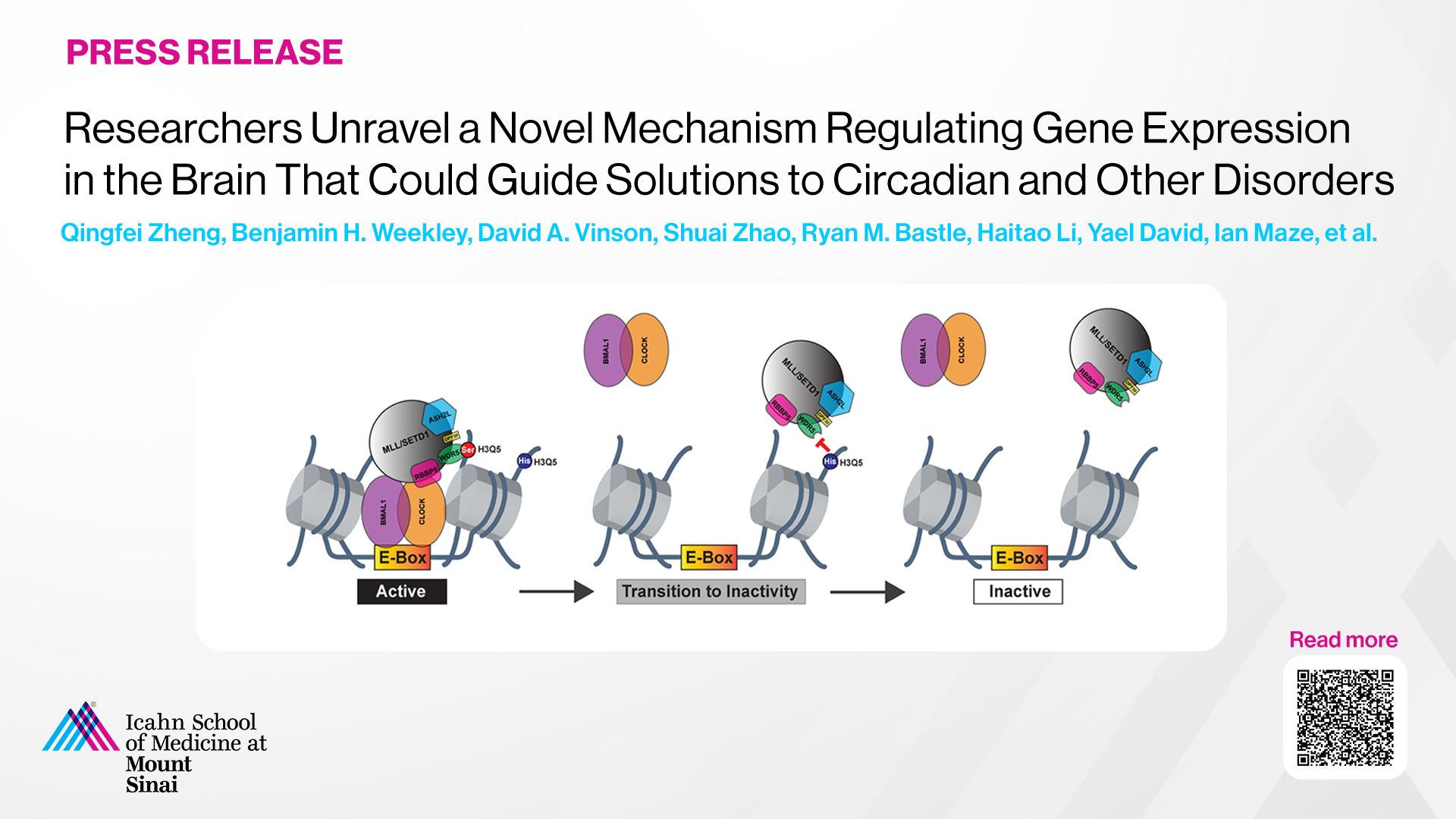
Bidirectional histone monoaminylation dynamics regulate neural rhythmicity
Qingfei Zheng, Benjamin H. Weekley, David A. Vinson, Shuai Zhao, Ryan M. Bastle, Robert E. Thompson, Stephanie Stransky, Aarthi Ramakrishnan, Ashley M. Cunningham, Sohini Dutta, Jennifer C. Chan, Giuseppina Di Salvo, Min Chen, Nan Zhang, Jinghua Wu, Sasha L. Fulton, Lingchun Kong, Haifeng Wang, Baichao Zhang, Lauren Vostal, Akhil Upad, Lauren Dierdorff, Li Shen, Henrik Molina, Simone Sidoli, Tom W. Muir, Haitao Li, Yael David & Ian Maze
Nature. 2025 Jan 8. doi: 10.1038/s41586-024-08371-3.
2024
Hippocampal γCaMKII dopaminylation promotes synaptic-to-nuclear signaling and memory formation
Andrew F. Stewart, Sasha L. Fulton, Romain Durand-de Cuttoli, Robert E. Thompson, Peng-Jen Chen, Elizabeth Brindley, Bulent Cetin, Lorna A. Farrelly, Rita Futamura, Sarah Claypool, Ryan M. Bastle, Giuseppina Di Salvo, Christopher Peralta, Henrik Molina, Erdene Baljinnyam, Samuele G. Marro, Scott J. Russo, Robert J. DeVita, Tom W. Muir, Ian Maze
https://doi.org/10.1101/2024.09.19.613951
A Novel Role for the Histone Demethylase JMJD3 in Mediating Heroin-Induced Relapse-Like Behaviors
Swarup Mitra, Craig T Werner, Treefa Shwani, Ana Garcia Lopez, Dale Federico, Kate Higdon, Xiaofang Li, Pedro H Gobira, Shruthi A Thomas, Jennifer A Martin, Chunna An, Ramesh Chandra, Ian Maze, Rachel Neve, Mary Kay Lobo, Amy M Gancarz, David M Dietz
Biol Psychiatry. 2024 Jul 15:S0006-3223(24)01452-5. doi: 10.1016/j.biopsych.2024.06.028.
Serotonin Transporter-dependent Histone Serotonylation in Placenta Contributes to the Neurodevelopmental Transcriptome
Jennifer C Chan , Natalia Alenina , Ashley M Cunningham , Aarthi Ramakrishnan , Li Shen , Michael Bader , Ian Maze ;
J Mol Biol 2024 Apr 1;2024.01.23 436(7):168454. doi: 10.10167
Repurposing the dopamine transporter antagonist vanoxerine to treat colorectal cancer
Winnie Chen, Ian Maze
Nat Cancer. 2024 Mar 5;2024.03.05. (3):378-379. doi: 10.1038
Early-life stress alters chromatin modifications in VTA to prime stress sensitivity
Luke T Geiger, Julie-Anne Balouek, Lorna A Farrelly, Andy S Chen, Megan Tang, Shannon N Bennett, Eric J Nestler, Benjamin A Garcia, Ian Maze, Catherine Jensen Peña
bioRxiv 2024 Mar 14;2024.03.14.584631. doi: 10.1101/2024.03.14.584631.
Bioorthogonal labeling and enrichment of histone monoaminylation reveal its accumulation and regulatory function in cancer cell chromatin
Nan Zhang, Jinghua Wu, Farzana Hossain, Haidong Peng, Huapeng Li, Connor Gibson, Min Chen, Huan Zhang, Shuaixin Gao, Xinru Zheng, Yongdong Wang, Jiangjiang Zhu, Jing J Wang, Ian Maze, Qingfei Zheng
bioRxiv. 2024 Mar 26;2024.03.20.586010. doi: 10.1101/2024.03.20.586010. Preprint
2023
Histone H3 serotonylation dynamics in dorsal raphe nucleus contribute to stress- and antidepressant-mediated gene expression and behavior
Amni Al-Kachak, Sasha L. Fulton, Giuseppina Di Salvo, Jennifer C. Chan, Lorna A. Farrelly, Ashley E. Lepack, Ryan M. Bastle, Lingchun Kong, Flurin Cathomas, Emily L. Newman, Caroline Menard, Aarthi Ramakrishnan, Polina Safovich, Yang Lyu, Herbert E. Covington 3rd, Li Shen, Kelly Gleason, Carol A. Tamminga, Scott J. Russo, Ian Maze
bioRxiv. 2023 Sep 16;2023.05.04.539464 doi: 10.1101/2023.05.04.539464
Post-translational modifications of histone proteins by monoamine neurotransmitters
Amni Al-Kachak, Ian Maze
Curr Opin Chem Biol. 2023 Apr 11;74:102302. doi: 10.1016/j.cbpa.2023.102302.
Flexible and site-specific manipulation of histones in live animals
Efrat Finkin-Groner, Amni Al-Kachak, Albert Agustinus, Ryan Bastle, Ashley Lepack, Yang Lyu, Ian Maze, Yael David
bioRxiv. 2023 Mar 20;2023.03.19.533378. doi: 10.1101/2023.03.19.533378. Preprint
Dissociable contributions of the amygdala and ventral hippocampus to stress-induced changes in defensive behavior
Zachary T Pennington, Alexa R LaBanca, Patlapa Sompolpong, Zoe Christenson Wick, Yu Feng, Zhe Dong, Taylor R Francisco, Lingxuan Chen, Sasha L Fulton, Ian Maze, Tristan Shuman, Denise J Cai
bioRxiv. 2023 Mar 11;2023.02.27.530077. doi: 10.1101/2023.02.27.530077. Preprint
Histone H3 dopaminylation in nucleus accumbens, but not medial prefrontal cortex, contributes to cocaine-seeking following prolonged abstinence
Ian Maze, Andrew F Stewart, Ashley L Lepack, Sasha L Fulton, Polina Safovich
Mol Cell Neurosci. 2023 Feb 24;103824. doi: 10.1016/j.mcn.2023.103824.
2022
Histone monoaminylation dynamics are regulated by a single enzyme and promote neural rhythmicity
Qingfei Zheng, Ryan M. Bastle, Shuai Zhao, Lingchun Kong, Lauren Vostal, Aarthi Ramakrishnan, Li Shen, Sasha L. Fulton, Haifeng Wang, Baichao Zhang, Akhil Upad, Lauren Dierdorff, Robert E. Thompson, Henrik Molina, Stephanie Stransky, Simone Sidoli, Tom W. Muir, Haitao Li, Yael David, Ian Maze
bioRxiv. 2022 Dec 6; doi: 10.1101/2022.12.06.519310
Rescue of deficits by Brwd1 copy number restoration in the Ts65Dn mouse model of Down syndrome
Sasha L Fulton*, Wendy Wenderski*, Ashley E Lepack*, Andrew L Eagle, Tomas Fanutza, Ryan M Bastle, Aarthi Ramakrishnan, Emma C Hays, Arianna Neal, Jaroslav Bendl, Lorna A Farrelly, Amni Al-Kachak, Yang Lyu, Bulent Cetin, Jennifer C Chan, Tina N Tran, Rachael L Neve, Randall J Roper, Kristen J Brennand, Panos Roussos, John C Schimenti, Allyson K Friedman, Li Shen, Robert D Blitzer, Alfred J Robison, Gerald R Crabtree, Ian Maze
Nat Commun. 2022 Oct 26;13(1):6384. doi: 10.1038/s41467-022-34200-0
TGM2-mediated histone transglutamination is dictated by steric accessibility
Bradley J. Lukasak, Michelle M. Mitchener, Lingchun Kong, Barbara E. Dul, Cole D. Lazarus, Aarthi Ramakrishnan, Jizhi Ni, Li Shen, Ian Maze, and Tom W. Muir
Proc Natl Acad Sci USA. 2022 Oct 25;119(43):e2208672119. doi: 10.1073/pnas.2208672119
Sex-biased effects on hippocampal circuit development by perinatal SERT expression in CA3 pyramidal neurons
Roberto De Gregorio, Galadu Subah, Jennifer C Chan, Luisa Speranza, Xiaolei Zhang, Aarthi Ramakrishnan, Li Shen, Ian Maze, Patric K Stanton, Ji Y Sze
Development. 2022 Sep 30. doi: 10.1242/dev.200549
Chromatin profiling in human neurons reveals aberrant roles for histone acetylation and BET family proteins in schizophrenia
Lorna A Farrelly*, Shuangping Zheng*, Nadine Schrode, Aaron Topol, Natarajan V Bhanu, Ryan M Bastle, Aarthi Ramakrishnan, Jennifer C Chan, Bulent Cetin, Erin Flaherty, Li Shen, Kelly Gleason, Carol A Tamminga, Benjamin A Garcia, Haitao Li, Kristen J Brennand, Ian Maze
Nat Commun. 2022 Apr 22;13(1):2195. doi: 10.1038/s41467-022-29922-0
Histone H3 dopaminylation in ventral tegmental area underlies heroin-induced transcriptional and behavioral plasticity in male rats
Sasha L Fulton*, Swarup Mitra* Ashley E Lepack, Jennifer A Martin, Andrew F Stewart, Jacob Converse, Mason Hochstetler, David M Dietz, Ian Maze
Neuropsychopharmacology. 2022 Jan 29. doi: 10.1038/s41386-022-01279-4
2021
Histone H3Q5 serotonylation stabilizes H3K4 methylation and potentiates its readout
Shuai Zhao, Kelly N. Chuh, Baichao Zhang, Barbara E. Dul, Robert E. Thompson, Lorna A. Farrelly, Xiaohui Liu, Ning Xu, Yi Xue, Robert G. Roeder, Ian Maze, Tom W. Muir, and Haitao Li
PNAS. 2021 Feb 9;118(6):e2016742118. doi: 10.1073/pnas.2016742118
Extracellular histones, a new class of inhibitory molecules of CNS axonal regeneration
Mustafa M Siddiq, Sari S Hannila, Yana Zorina, Elena Nikulina, Vera Rabinovich, Jianwei Hou, Rumana Huq, Erica L Richman, Rosa E Tolentino, Jens Hansen, Adam Velenosi, Brian K Kwon, Stella E Tsirka, Ian Maze, Robert Sebra, Kristin G Beaumont, Carlos A Toro, Christopher P Cardozo, Ravi Iyengar, Marie T Filbin
Brain Commun. 2021 Nov 13;3(4):fcab271. doi: 10.1093/braincomms/fcab271
2020
Circuit-specific hippocampal ΔFosB underlies resilience to stress-induced social avoidance
Andrew L Eagle, Claire E Manning, Elizabeth S Williams, Ryan M Bastle, Paula A Gajewski, Amber Garrison, Alexis J Wirtz, Seda Akguen, Katie Brandel-Ankrapp, Wilson Endege, Frederick M Boyce, Yoshinori N Ohnishi, Michelle Mazei-Robison, Ian Maze, Rachel L Neve, Alfred J Robison
Nat Commun. 2020 Sep 8;11(1):4484. doi: 10.1038/s41467-020-17825-x.
Epigenetics of Drug Addiction
Andrew F. Stewart, Sasha L. Fulton and Ian Maze
Cold Spring Harb Perspect Med. 2020 Jun 8:a040253. doi: 10.1101/cshperspect.a040253. | Download reprint
Nothing Is Yet Set in (Hi)stone: Novel Post-Translational Modifications Regulating Chromatin Function
Jennifer C Chan, Ian Maze
Trends Biochem Sci. 2020 Jun 1. Online ahead of print. DOI: 10.1016/j.tibs.2020.05.009
Dopaminylation of histone H3 in ventral tegmental area regulates cocaine seeking.
Ashley E. Lepack, Craig T. Werner, Andrew F. Stewart, Sasha L. Fulton, Ping Zhong, Lorna A. Farrelly, Alexander C. W. Smith, Aarthi Ramakrishnan, Yang Lyu, Ryan M. Bastle, Jennifer A. Martin, Swarup Mitra, Richard M. O’Connor, Zi-Jun Wang, Henrik Molina, Gustavo Turecki, Li Shen, Zhen Yan, Erin S. Calipari, David M. Dietz, Paul J. Kenny and Ian Maze
Science 10 Apr 2020, Vol. 368, Issue 6487, pp. 197-201, DOI: 10.1126/science.aaw8806 | Download reprint
2019
Ubiquitin-proteasomal regulation of chromatin remodeler INO80 in the nucleus accumbens mediates persistent cocaine craving.
Werner CT, Mitra S, Martin JA, Stewart AF, Lepack AE, Ramakrishnan A, Gobira PH, Wang ZJ, Neve RL, Gancarz AM, Shen L, Maze I, Dietz DM.
Sci Adv. 2019 Oct 9;5(10):eaay0351. doi: 10.1126/sciadv.aay0351. eCollection 2019 Oct.
Translational Molecular Approaches in Substance Abuse Research.
Fulton SL, Maze I.
Handb Exp Pharmacol. 2019 Oct 19. doi: 10.1007/164_2019_259.
Epigenetic regulation of hippocampal FosB expression controls behavioral responses to cocaine.
Gajewski PA, Eagle AL, Williams ES, Manning CE, Lynch H, McCornack C, Maze I, Heller EA, Robison AJ.
J Neurosci. 2019 Sep 2. pii: 0800-19.
Stress resilience is promoted by a Zfp189-driven transcriptional network in prefrontal cortex.
Lorsch ZS, Hamilton PJ, Ramakrishnan A, Parise EM, Salery M, Wright WJ, Lepack AE, Mews P, Issler O, McKenzie A, Zhou X, Parise LF, Pirpinias ST, Ortiz Torres I, Kronman HG, Montgomery SE, Loh YE, Labonté B, Conkey A, Symonds AE, Neve RL, Turecki G, Maze I, Dong Y, Zhang B, Shen L, Bagot RC, Nestler EJ.
Nat Neurosci. 2019 Sep;22(9):1413-1423
An emerging perspective on ‘histone code’ mediated regulation of neural plasticity and disease.
Farrelly LA, Maze I.
Curr Opin Neurobiol. 2019 Aug 1;59:157-163.
Histone Crotonylation Makes Its Mark in Depression Research.
Chan JC, Maze I.
Biol Psychiatry. 2019 Apr 15;85(8):616-618.
Histone serotonylation is a permissive modification that enhances TFIID binding to H3K4me3
Farrelly LA, Thompson RE, Zhao S, Lepack AE, Lyu Y, Bhanu NV, Zhang B, Loh YHE, Ramakrishnan A, Vadodaria KC, Heard KJ, Erikson G, Nakadai T, Bastle RM, Lukasak BJ, Zebroski H, Alenina N, Bader M, Berton O, Roeder RG, Molina H, Gage FH, Shen L, Garcia BA, Li H, Muir TW and Maze I
Nature 2019 March doi.org/10.1038/s41586-019-1024-7
Chromatin regulation in complex brain disorder
Bastle RM, Maze I
Current Opinion in Behavioral Sciences 2019, 25:57-65
Knockdown of the histone di-methyltransferase G9a in nucleus accumbens shell decreases cocaine self-administration, stress-induced reinstatement, and anxiety.
Anderson EM, Sun H, Guzman D, Taniguchi M, Cowan CW, Maze I, Nestler EJ, Self DW.
Neuropsychopharmacology. 2018 Dec 26. doi: 10.1038/s41386-018-0305-4. [Epub ahead of print]
2018
Cell-type-specific role for nucleus accumbens neuroligin-2 in depression and stress susceptibility
M Heshmati, H Aleyasin, C Menard, DJ Christoffel, ME Flanigan, ML Pfau, …
Proceedings of the National Academy of Sciences 115 (5), 1111-1116 — 2018
Activated Protein C blocks the inhibitory effect on neurite outgrowth by extracellular histones that mediates its inhibition through a retrograde YB-1 signal
MM Siddiq, SS Hannila, Y Zorina, E Nikulina, V Rabinovich, J Hou, R Huq, …
bioRxiv, 365825 — 2018
2017
Macromolecular isolation and purification in AMS
B Buchholz, K Spalding, R Dreier, I Maze, B Mailloux, S Carratt, …
ABSTRACTS OF PAPERS OF THE AMERICAN CHEMICAL SOCIETY 253 — 2017
2016 & Earlier
Aberrant H3.3 dynamics in NAc promote vulnerability to depressive-like behavior.
Lepack AE, Bagot RC, Peña CJ, Loh YE, Farrelly LA, Lu Y, Powell SK, Lorsch ZS, Issler O, Cates HM, Tamminga CA, Molina H, Shen L, Nestler EJ, Allis CD, Maze I.
Proc Natl Acad Sci U S A. 2016 Nov 1;113(44):12562-12567.
Histone arginine methylation in cocaine action in the nucleus accumbens.
Damez-Werno DM, Sun H, Scobie KN, Shao N, Rabkin J, Dias C, Calipari ES, Maze I, Pena CJ, Walker DM, Cahill ME, Chandra R, Gancarz A, Mouzon E, Landry JA, Cates H, Lobo MK, Dietz D, Allis CD, Guccione E, Turecki G, Defilippi P, Neve RL, Hurd YL, Shen L, Nestler EJ.
Proc Natl Acad Sci U S A. 2016 Aug 23;113(34):9623-8. doi: 10.1073/pnas.1605045113. Epub 2016 Aug 9.
Current Proteomic Methods to Investigate the Dynamics of Histone Turnover in the Central Nervous System.
Farrelly LA, Dill BD, Molina H, Birtwistle MR, Maze I.
Methods Enzymol. 2016;574:331-54. doi: 10.1016/bs.mie.2016.01.013. Epub 2016 Feb 16.
Circuit-wide Transcriptional Profiling Reveals Brain Region-Specific Gene Networks Regulating Depression Susceptibility.
Bagot RC, Cates HM, Purushothaman I, Lorsch ZS, Walker DM, Wang J, Huang X, Schlüter OM, Maze I, Peña CJ, Heller EA, Issler O, Wang M, Song WM, Stein JL, Liu X, Doyle MA, Scobie KN, Sun HS, Neve RL, Geschwind D, Dong Y, Shen L, Zhang B, Nestler EJ.
Neuron. 2016 Jun 1;90(5):969-83. doi: 10.1016/j.neuron.2016.04.015. Epub 2016 May 12.
Histone turnover and chromatin accessibility: Critical mediators of neurological development, plasticity, and disease.
Wenderski W, Maze I.
Bioessays. 2016 May;38(5):410-9. doi: 10.1002/bies.201500171. Epub 2016 Mar 15.
2015
ACF chromatin-remodeling complex mediates stress-induced depressive-like behavior.
Sun H, Damez-Werno DM, Scobie KN, Shao NY, Dias C, Rabkin J, Koo JW, Korb E, Bagot RC, Ahn FH, Cahill ME, Labonté B, Mouzon E, Heller EA, Cates H, Golden SA, Gleason K, Russo SJ, Andrews S, Neve R, Kennedy PJ, Maze I, Dietz DM, Allis CD, Turecki G, Varga-Weisz P, Tamminga C, Shen L, Nestler EJ.Nature Medicine. 2015 Oct;21(10):1146-53. doi: 10.1038/nm.3939. Epub 2015 Sep 21.
Critical role of histone turnover in neuronal transcription and plasticity
Maze I, Wenderski W, Noh KM, Bagot R, Tzavaras N, Purushothaman I, Elsässer S, Guo Y, Ionete C, Hurd Y, Tamminga C, Halene T, Farrelly L, Soshnev A, Wen D, Rafii S, Birtwistle M, Akbarian S, Buchholz B, Blitzer R, Nestler E, Yuan ZF, Garcia B, Shen L, Molina H, Allis CD
Neuron. 2015 Jul 1;87(1):77-94. doi: 10.1016/j.neuron.2015.06.014.
Ventral hippocampal afferents to the nucleus accumbens regulate susceptibility to depression.
Bagot RC, Parise EM, Peña CJ, Zhang HX, Maze I, Chaudhury D, Persaud B, Cachope R, Bolaños-Guzmán CA, Cheer J, Deisseroth K, Han MH, Nestler EJ.
Nature Communications. 2015 May 8;6:7062. doi: 10.1038/ncomms8062.
Antidepressant action of HDAC inhibition in the prefrontal cortex.
Covington HE 3rd, Maze I, Vialou V, Nestler EJ.
Neuroscience. 2015 Jul 9;298:329-35. doi: 10.1016/j.neuroscience.2015.04.030. Epub 2015 Apr 20.
2014
ATRX tolerates activity-dependent histone H3 methyl/phos switching to maintain repetitive element silencing in neurons.
Noh KM, Maze I, Zhao D, Xiang B, Wenderski W, Lewis PW, Shen L, Li H, Allis CD.
Proceedings of the National Academy of Sciences. 2015 Jun 2;112(22):6820-7. doi: 10.1073/pnas.1411258112. Epub 2014 Dec 23.
Analytical tools and current challenges in the modern era of neuroepigenomics.
Maze I, Shen L, Zhang B, Garcia BA, Shao N, Mitchell A, Sun H, Akbarian S, Allis CD, Nestler EJ.
Nature Neuroscience. 2014 Nov;17(11):1476-90. doi: 10.1038/nn.3816. Epub 2014 Oct 28. Review.
Chronic cocaine-regulated epigenomic changes in mouse nucleus accumbens.
Feng J, Wilkinson M, Liu X, Purushothaman I, Ferguson D, Vialou V, Maze I, Shao N, Kennedy P, Koo J, Dias C, Laitman B, Stockman V, LaPlant Q, Cahill ME, Nestler EJ, Shen L.
Genome Biology 2014 April 22; 15(4).
Every amino acid matters: essential contributions of histone variants to mammalian development and disease.
Maze I, Noh KM, Soshnev AA, Allis CD.
Nature Reviews. Genetics 2014 Apr; 15(4).
G9a influences neuronal subtype specification in striatum.
Maze I, Chaudhury D, Dietz DM, Von Schimmelmann M, Kennedy PJ, Lobo MK, Sillivan SE, Miller ML, Bagot RC, Sun H, Turecki G, Neve RL, Hurd YL, Shen L, Han MH, Schaefer A, Nestler EJ.
Nature Neuroscience 2014 Apr; 17(4).
ΔFosB induction in prefrontal cortex by antipsychotic drugs is associated with negative behavioral outcomes.
Dietz DM, Kennedy PJ, Sun H, Maze I, Gancarz AM, Vialou V, Koo JW, Mouzon E, Ghose S, Tamminga CA, Nestler EJ.
Neuropsychopharmacology 2014 Feb; 39(3).
2013
diffReps: detecting differential chromatin modification sites from ChIP-seq data with biological replicates.
Shen L, Shao NY, Liu X, Maze I, Feng J, Nestler EJ.
PloS one 2013; 8(6).
Class I HDAC inhibition blocks cocaine-induced plasticity by targeted changes in histone methylation.
Kennedy PJ, Feng J, Robison AJ, Maze I, Badimon A, Mouzon E, Chaudhury D, Damez-Werno DM, Haggarty SJ, Han MH, Bassel-Duby R, Olson EN, Nestler EJ.
Nature Neuroscience 2013 Apr; 16(4).
Histone regulation in the CNS: basic principles of epigenetic plasticity.
Maze I, Noh KM, Allis CD.
Neuropsychopharmacology : Official Publication of the American College of Neuropsychopharmacology 2013 Jan; 38(1).
2012
Morphine epigenomically regulates behavior through alterations in histone H3 lysine 9 dimethylation in the nucleus accumbens.
Sun H, Maze I, Dietz DM, Scobie KN, Kennedy PJ, Damez-Werno D, Neve RL, Zachariou V, Shen L, Nestler EJ.
The Journal of Neuroscience : the Official Journal of the Society for Neuroscience 2012 Nov; 32(48).
Rac1 is essential in cocaine-induced structural plasticity of nucleus accumbens neurons.
Dietz DM, Sun H, Lobo MK, Cahill ME, Chadwick B, Gao V, Koo JW, Mazei-Robison MS, Dias C, Maze I, Damez-Werno D, Dietz KC, Scobie KN, Ferguson D, Christoffel D, Ohnishi Y, Hodes GE, Zheng Y, Neve RL, Hahn KM, Russo SJ, Nestler EJ.
Nature Neuroscience 2012 Jun; 15(6).
2011
A role for repressive histone methylation in cocaine-induced vulnerability to stress.
Covington HE, Maze I, Sun H, Bomze HM, DeMaio KD, Wu EY, Dietz DM, Lobo MK, Ghose S, Mouzon E, Neve RL, Tamminga CA, Nestler EJ.
Neuron 2011 Aug; 71(4).
Behavioral epigenetics.
Lester BM, Tronick E, Nestler E, Abel T, Kosofsky B, Kuzawa CW, Marsit CJ, Maze I, Meaney MJ, Monteggia LM, Reul JM, Skuse DH, Sweatt JD, Wood MA.
Annals of the New York Academy of Sciences 2011 May; 1226.
Cocaine dynamically regulates heterochromatin and repetitive element unsilencing in nucleus accumbens.
Maze I, Feng J, Wilkinson MB, Sun H, Shen L, Nestler EJ.
Proceedings of the National Academy of Sciences of the United States of America 2011 Feb; 108(7).
The epigenetic landscape of addiction.
Maze I, Nestler EJ.
Annals of the New York Academy of Sciences 2011 Jan; 1216.
2010
Antidepressant effect of optogenetic stimulation of the medial prefrontal cortex.
Covington HE, Lobo MK, Maze I, Vialou V, Hyman JM, Zaman S, LaPlant Q, Mouzon E, Ghose S, Tamminga CA, Neve RL, Deisseroth K, Nestler EJ.
The Journal of Neuroscience : the Official Journal of the Society for Neuroscience 2010 Dec; 30(48).
Serum response factor promotes resilience to chronic social stress through the induction of DeltaFosB.
Vialou V, Maze I, Renthal W, LaPlant QC, Watts EL, Mouzon E, Ghose S, Tamminga CA, Nestler EJ.
The Journal of Neuroscience : the Official Journal of the Society for Neuroscience 2010 Oct; 30(43).
Dnmt3a regulates emotional behavior and spine plasticity in the nucleus accumbens.LaPlant Q, Vialou V, Covington HE, Dumitriu D, Feng J, Warren BL, Maze I, Dietz DM, Watts EL, Iñiguez SD, Koo JW, Mouzon E, Renthal W, Hollis F, Wang H, Noonan MA, Ren Y, Eisch AJ, Bolaños CA, Kabbaj M, Xiao G, Neve RL, Hurd YL, Oosting RS, Fan G, Morrison JH, Nestler EJ.
Nature Neuroscience 2010 Sep; 13(9).
Transcriptional mechanisms: underlying addiction-related structural plasticity.
Maze I, Russo SJ.
Molecular Interventions 2010 Aug; 10(4).
Essential role of the histone methyltransferase G9a in cocaine-induced plasticity.
Maze I, Covington HE, Dietz DM, LaPlant Q, Renthal W, Russo SJ, Mechanic M, Mouzon E, Neve RL, Haggarty SJ, Ren Y, Sampath SC, Hurd YL, Greengard P, Tarakhovsky A, Schaefer A, Nestler EJ.
Science (New York, N.Y.) 2010 Jan; 327(5962).
2009
Antidepressant actions of histone deacetylase inhibitors.
Covington HE, Maze I, LaPlant QC, Vialou VF, Ohnishi YN, Berton O, Fass DM, Renthal W, Rush AJ, Wu EY, Ghose S, Krishnan V, Russo SJ, Tamminga C, Haggarty SJ, Nestler EJ.
The Journal of Neuroscience : the Official Journal of the Society for Neuroscience 2009 Sep; 29(37).
Genome-wide analysis of chromatin regulation by cocaine reveals a role for sirtuins.
Renthal W, Kumar A, Xiao G, Wilkinson M, Covington HE, Maze I, Sikder D, Robison AJ, LaPlant Q, Dietz DM, Russo SJ, Vialou V, Chakravarty S, Kodadek TJ, Stack A, Kabbaj M, Nestler EJ.
Neuron 2009 May; 62(3).
Role of nuclear factor kappaB in ovarian hormone-mediated stress hypersensitivity in female mice.
LaPlant Q, Chakravarty S, Vialou V, Mukherjee S, Koo JW, Kalahasti G, Bradbury KR, Taylor SV, Maze I, Kumar A, Graham A, Birnbaum SG, Krishnan V, Truong HT, Neve RL, Nestler EJ, Russo SJ.
Biological Psychiatry 2009 May; 65(10).
Nuclear factor kappa B signaling regulates neuronal morphology and cocaine reward.
Russo SJ, Wilkinson MB, Mazei-Robison MS, Dietz DM, Maze I, Krishnan V, Renthal W, Graham A, Birnbaum SG, Green TA, Robison B, Lesselyong A, Perrotti LI, Bolaños CA, Kumar A, Clark MS, Neumaier JF, Neve RL, Bhakar AL, Barker PA, Nestler EJ.
The Journal of Neuroscience : the Official Journal of the Society for Neuroscience 2009 Mar; 29(11).
2008
Delta FosB mediates epigenetic desensitization of the c-fos gene after chronic amphetamine exposure.
Renthal W, Carle TL, Maze I, Covington HE, Truong HT, Alibhai I, Kumar A, Montgomery RL, Olson EN, Nestler EJ.
The Journal of Neuroscience : the Official Journal of the Society for Neuroscience 2008 Jul; 28(29).
Distinct patterns of DeltaFosB induction in brain by drugs of abuse.
Perrotti LI, Weaver RR, Robison B, Renthal W, Maze I, Yazdani S, Elmore RG, Knapp DJ, Selley DE, Martin BR, Sim-Selley L, Bachtell RK, Self DW, Nestler EJ.
Synapse (New York, N.Y.) 2008 May; 62(5).
2007
Histone deacetylase 5 epigenetically controls behavioral adaptations to chronic emotional stimuli.
Renthal W, Maze I, Krishnan V, Covington HE, Xiao G, Kumar A, Russo SJ, Graham A, Tsankova N, Kippin TE, Kerstetter KA, Neve RL, Haggarty SJ, McKinsey TA, Bassel-Duby R, Olson EN, Nestler EJ.
Neuron 2007 Nov; 56(3).
Acute ethanol ingestion produces dose-dependent effects on motor behavior in the honey bee (Apis mellifera).
Maze IS, Wright GA, Mustard JA.
Journal of Insect Physiology; 52(11-12).
Book Chapters
Book Chapters
Wenderski W, Maze I. (2014). Epigenetic mechanisms of drug addiction vulnerability. In J Peedicayil, D Avramopoulos and D Grayson (Eds.), Epigenetics in Psychiatry. Elsevier, New York, p. 441-462.
Maze I, Nestler EJ. (2012), Epigenetics and drug addiction. In Sassone-Corsi P. and Christen Y. (Eds.), Epigenetics, Brain and Behavior. Springer-Verlag, Heidelberg, p. 145-160.
Featured
Meet the Team












Previous Lab Members

Newaz Ahmed, Ph.D.
Associate Scientist at SingulOmics

Amni Al-Kachak, PhD
Senior Medical Partner at Flywheel Partners

Ryan Bastle, Ph.D.
Medical Science Liaison at AbbVie

Lizzie Brindley, Ph.D.
Scientist at Johnson & Johnson

Sohini Dutta, Ph.D.
Consultant at Trinity Life Sciences

Marisa Cerio
Associate Director, Grants Office, NY Stem Cell Foundation

Winnie Chen
MD/PhD Student

Lorna Farrelly, Ph.D.
Scientist at Regeneron

Sasha Fulton, PhD
Postdoctoral fellow at Columbia University

Sydney Grossman
Volunteer

Lingchun Kong, Ph.D.
Director, Active Motif Shanghai


Yang Lyu
Senior Associate Researcher

Polina Safovich
Medical Scribe at CityMD

Andrew Stewart
Postdoctoral fellow at Stanford University

Cristina Toro
Volunteer

Xiaoran Wei, Ph.D.
Postdoctoral Fellow
Grants
Active Grants
AGR-25721 (Ian Maze)
Ono Pharmaceutical
Molecular control of inflammatory mechanisms contributing to major depressive disorder
The major goals of this project are to examine novel roles for inflammatory signaling in OFC, mPFC and NAc in the regulation of aberrant glial-to-neuronal communication and stress vulnerability related to major depressive disorder and other mood-related syndromes.
R01 DA056595 (Ian Maze, David Dietz)
National Institute on Drug Abuse (NIH/NIDA)
Contributions of aberrant synaptic protein monoaminylation to opiate use disorder
The major goals of this project are to examine novel roles for synaptic dopaminylation in NAc in the precipitation of aberrant synapse-to-nucleus signaling and relapse vulnerability during periods of withdrawal from chronic heroin exposures.
R01 MH116900, (Maze)
National Institutes of Health/NIH/DHHS (NIMH)
Molecular studies of neural histone monoaminylation in normal and aberrant brain plasticity
The major goals of this project are to investigate cell-type, brain region and sex specific roles for histone monoaminylation in the precipitation of depressive-like behaviors and antidepressant actions in rodent models of major depressive disorder.
R01 HD097088 (Maze)
National Institutes of Health/NIH/DHHS (NICHD)
Aberrant chromatin regulatory mechanisms in Down syndrome brain
The major goals of this project are to investigate potential roles for the chromatin “reader” protein BRWD1 in Down syndrome associated neurological and cognitive deficits, and more broadly to examine epigenetic dysregulation in Trisomy 21
2022 Neurobiology of Brain Disorders Award (Shared with Jeremy Day, Ph.D.)
The McKnight Endowment Fund for Neuroscience
Leveraging single-cell epigenomics for targeted manipulation of drug-activated ensembles: Drs. Day and Maze are researching the epigenetic underpinnings of addiction, identifying neural ensembles that have been hijacked by drug exposures, thereby disposing individuals to relapse.
R01 DA056592 (Drew Kiraly; Co-I Maze)
National Institute on Drug Abuse (NIH/NIDA)
Targeting Gut-brain signaling to reduce cocaine-seeking behaviors
Past Grants
DP1 DA042078, Avenir Award (Maze)
National Institutes of Health/NIH/DHHS (NIDA)
Roles for histone monoaminylation in cocaine-induced transcriptional and behavioral plasticity
The major goals of this project are to understand whether dopaminergic dysfunction in rodent models of cocaine addiction impact the expression of histone dopaminylation in brain, as well as how such alterations in monoaminylation impact addictive-like phenotypes.
P50 MH096890, Conte Center (Nestler)
National Institutes of Health/NIH/DHHS (NIMH)
Epigenetic mechanisms in depression
The major goals of this project are to continue investigating the “epigenetic” mechanisms underlying human
major depressive disorder, as well as individual responses to antidepressants, using both rodent model systems and human postmortem tissues.
Role: Co-PI (Project 3)
One Mind – Early Diagnostics Rising Star Research Award (Maze)
One Mind Institute
Validation of histone serotonylation as a predictive blood biomarker for stress susceptibility, major
depressive disorder and antidepressant efficacy
The overarching goal of this project is to fully test the hypothesis that newly discovered histone serotonylation may be used as a peripheral biomarker for MDD and/or antidepressant efficacy in human patients in order to aid in the implementation of more effective therapeutic interventions.
R21 DA044767, (Maze/David Shternberg)
National Institutes of Health/NIH/DHHS (NIDA)
Application of intein-based chemical methods to directly manipulate neuronal histone modifications in rodent models of addiction
The major goals of this project are to further develop and implement novel intein-based chemical
methodologies for directly manipulating histone and other protein modifications in vivo in brain, with an extended emphasis on applying these approaches for use in rodent addiction models.
Basil O’Connor Starter Scholar Research Award (Maze)
March of Dimes
Aberrant epigenetic mechanisms contributing to Down syndrome: a novel role for the histone reader
protein BRWD1
The major goals of this project are to characterize the role of BRWD1 and its target genes in mediating intellectual disability in Down syndrome brain.
Alfred P. Sloan Research Fellow in Neuroscience (Maze)
Alfred P. Sloan Foundation
Mechanistic Neuroepigenetics
This is a career award to promote my laboratory’s advancement of the new field of mechanistic
neuroepigenetics.
MQ15FIP100011 (Maze)
MQ: Transforming Mental Health Research Fellowship
Wellcome Trust
Histone monoaminylation in the CNS: novel insights into the cause and treatment of human depression
The major goals of this project are to understand whether serotonergic dysfunction in rodent models of human depression impact the expression of histone serotonylation in brain, as well as if current antidepressants affect such expression.
Fay/Frank Seed Grant (Maze)
Brain Research Foundation
Beyond neurotransmission: exploring roles for synaptic dopaminylation in drug-induced plasticity
The major goals of this project are to use novel chemical approaches to identify synaptic proteins that are dopaminylated in the context of drug abuse (specifically cocaine) for future molecular and behavioral characterizations.
NARSAD Young Investigator Award (Maze)
Brain & Behavior Research Foundation
Epigenomic profile and function of histone variant H3.3 dynamics in depression
The major goal of this project was to investigate the role of histone turnover/H3.3 dynamics in the mediation of
transcriptional abnormalities contributing to depressive-like behaviors in rodent models of human depression.
Rosen Family Research Scholar Award (Maze)
Friedman Brain Institute Pilot Fund Partnership
Function of a novel ‘reader’ of neuronal chromatin in Trisomy 21 associated Alzheimer’s disease
The major goals of this project are to develop a novel hiPSC derived neuronal model of Down syndrome in order
to investigate whether triplication of the neuronally enriched histone reader protein BRWD1 contributes to
observed neurological/synaptic impairments.
Active Grants (Lab Members)
F30 AG090096 (Chen, Winnie)
NIH National Institute on Aging (NIA)
Investigating Histone 3 Dopaminylation in Parkinson’s Disease
F31 NS132558 (Cunningham)
NIH National Research Service Award (NINDS)
Functional Role for H3 Serotonylation During Critical Periods of Postnatal Brain Development and Plasticity
Past Grants (Lab Members)
F32 1F32MH126534 (Chan)
National Institute of Mental Health (NIMH)
Impact of histone serotonylation domain organization on neurodevelopment
F31 DA055462 (Stewart)
NIH National Research Service Award (NRSA)
Identification of CaMKII as a novel substrate of dopaminylation following heroin self administration
F99 F99NS125774 (Fulton)
NIH National Institute of Neurological Disorders and Stroke (NINDS)
Characterizing effects of OFC astrocyte plasticity in inflammation response and stress vulnerability with single-cell resolution
F31 MH124425 (Fulton)
National Institutes of Health (NIMH)
Investigating Chromatin-Based Mechanisms Of Astrocyte Pathology During Inflammation Response And Stress-Induced Behaviors
NSF Graduate Research Fellowship (Stewart)
National Science Foundation/GRFP
It is awarded based upon an individual’s potential to “contribute to the US science and engineering enterprise.”
K99 MH120334 01 (Farrelly)
National Institutes of Health/RTCD (NIMH)
Studies of histone serotonylation and its role in transcriptional and neural plasticity
F31 DA045428 (Lepack)
National Institutes of Health/NIH/DHHS (NIDA)
Functions for histone dopaminylation in cocaine-induced plasticity
The major goals of this fellowship project are to understand whether dopaminergic dysfunction in rodent
models of cocaine addiction impact the expression of histone dopaminylation in brain, as well as how such
alterations in monoaminylation impact addictive-like phenotypes.
NARSAD Young Investigator Award (Farrelly)
Brain & Behavior Research Foundation
Histone serotonylation: a novel epigenetic mechanism mediating depressive-like phenotypes
The major goals of this young investigator award are to understand whether serotonergic dysfunction in
rodent models of human depression impact the expression of histone serotonylation in brain, as well as if
current antidepressants affect such expression.
T32 DA007135 (Devi)
National Institutes of Health/NIH/DHHS (NIDA)
Role of protein dopaminylation in drug addiction
The major goals of this project are to use novel chemical approaches to identify synaptic proteins that
are dopaminylated in the context of drug abuse (specifically cocaine and heroin) for future molecular
and behavioral characterizations.
Role: Postdoctoral Trainee (Bastle)
T32 MH087004 (Salton)
National Institutes of Health/NIH/DHHS (NIMH)
Genome-wide chromatin accessibility profiling in major depressive disorder reveals astrocytic specific
signatures of reward processing deficits in the orbitofrontal cortex
The major goals of this training grant project are to investigate epigenetic mechanisms underlying human
major depressive disorder, with a specific emphasis on chromatin mediated glial dysfunction leading to
reward processing deficits in the frontal cortex.
Role: Predoctoral Trainee (Fulton)
Awards
Ian Maze: 2023 Ian Maze: 2023 Blavatnik National Awards for Young Scientists Finalist (Life Sciences)
Ashley Cunningham: 2022 SfN Neuroscience Scholars Award (SfN)
Sasha Fulton: 2022 Winner of the NIH Outstanding Scholars in Neuroscience Award (OSNAP)
Jennifer Chan: 2022 Winner of the Robin Chemers Neustein Postdoctoral Fellowship Award
Ashley Lepack: 2020 Winner of the Nemko Prize in Cellular and Molecular Neuroscience
Ian Maze: 2019 Winner of Presidential Early Career Award for Scientists and Engineers (PECASE)
Ian Maze: 2019 Dr. Harold and Golden Laport Basic Research Award
Lorna Farrelly: The Friedman Brain Institute 2019 POSTDOC Award: for the best record of achievement in neuroscience.
Amni Al-Kachak: The Friedman Brain Institute 2019 Best Graduate Student Poster Award
Lorna Farrelly: Robin Chemers Neustein Award (The Robin Chemers Neustein Postdoctoral Fellowship award provides an exceptional female, early-career postdoctoral fellow with $25,000 to supplement that postdoctoral fellow’s current stipend/salary for one year.)
Ashley Lepack: 2018 Hausfeld Award (recognizing the most outstanding Neuroscience PhD student at the Icahn School of Medicine at Mount Sinai)
Ashley Lepack: Best Speaker Prize, Icahn School of Medicine at Mount Sinai’s Annual Neuroscience Retreat (2018)
Sasha Fulton: 2018 BRAIN Award (recognizing the most outstanding 2nd year Neuroscience PhD student at the Icahn School of Medicine at Mount Sinai)
Lorna Farrelly: Invited Speaker for the SPiNES Postdoctoral Seminar Series (Neuroscience Institute, New York University School of Medicine)
Lorna Farrelly: Best Poster Prize, Icahn School of Medicine at Mount Sinai’s Annual Neuroscience Retreat (2017)
Lorna Farrelly: 2nd Best Poster Prize, Icahn School of Medicine at Mount Sinai’s Annual Neuroscience Retreat (2016)
Ian Maze: ACNP Travel Award Winner (2015)
Ashley Lepack: Janelia Research Campus Conference Travel Award Winner (2015), “Behavioral Epigenetics: Conserved Mechanisms in Diverse Model Systems