Research

Brain tumors remain a deadly form of cancer. Our research goal is to provide a better understanding of the molecular machinery that drives proliferation, invasiveness, and therapy resistance of brain tumor cells. We aim to provide insights that will allow development of new targeted therapies against brain tumor cells.
Our research team focuses on the question of how axon guidance molecules regulate migration and proliferation of brain tumor cells. We are also investigating the molecular characteristics that define brain tumor stem cells by using molecular reporters.
Projects
Tumors of the brain are amongst the most lethal types of cancer. They account for over 175,000 cases per year worldwide, claiming the lives of thousands of patients (WHO World Cancer Report). Despite significant progress in the identification of genes involved in tumorigenesis, the etiology of brain tumors is still largely unknown, and incidence and mortality rates of these cancers have changed little over the past decade. The goal of our research is to identify the genetic factors that contribute to the formation and spreading of brain tumors. The identification of novel diagnostic markers and therapeutic targets will lead to better treatment strategies for this devastating disease.
Axon guidance molecules control cellular dynamics and malignancy of glioma cells
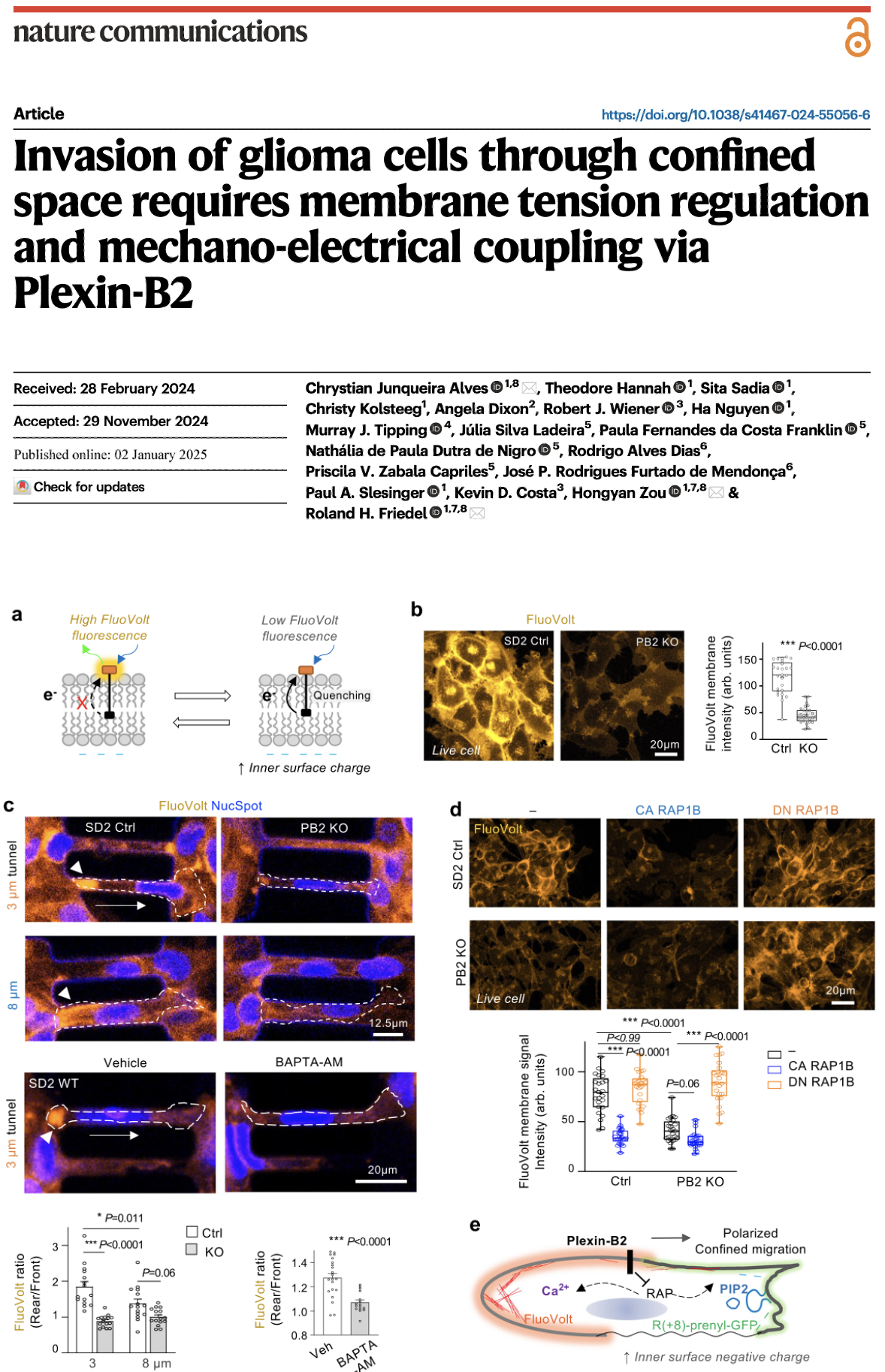
To identify novel molecules that regulate the invasive spreading behavior of brain tumor cells, we are performing functional genetic analyses with human glioma stem cell lines in vitro and in transplantation assays in vivo. We are focusing our studies on candidate genes of the axon guidance families and their downstream signaling components. A particular effort is on elucidating the role of Plexin-B2, an axon guidance receptor that plays an important role in formation of the developing nervous system, and which is highly expressed in malignant glioblastoma.
Cellular and molecular characteristics of glioblastoma stem cells
Brain tumor stem cells are believed to be a special subpopulation that exhibits enhanced tumorigenic capacity and resistance to conventional therapy. To identify underlying molecular mechanisms that control brain tumor stem cells in their migratory, self-renewal, and proliferative behaviors, we have engineered glioma cells with a cell division marker H2B-GFP. Cellular and molecular characteristics of cells with different cell division history are studied in orthotopic transplant models.
Publications
Featured
Meet the Team




Jesse Fisher
Volunteer